Categories of Acids and Bases

Before we dive in, be sure to download my FREE acid/base cheat sheet to follow along now and take with you to study on the go – Organic Chemistry Acid Base Cheat Sheet
Acids and bases will fall under one or more of the following three categories:
1. Arrhenius acids/bases
2. Bronsted-Lowry acids/bases
3. Lewis acids/bases
The key here is to recognize that while each classification has a specific definition, any given molecule can fall into more than one category, some into all 3. Again, something we’ll look at later in this article.
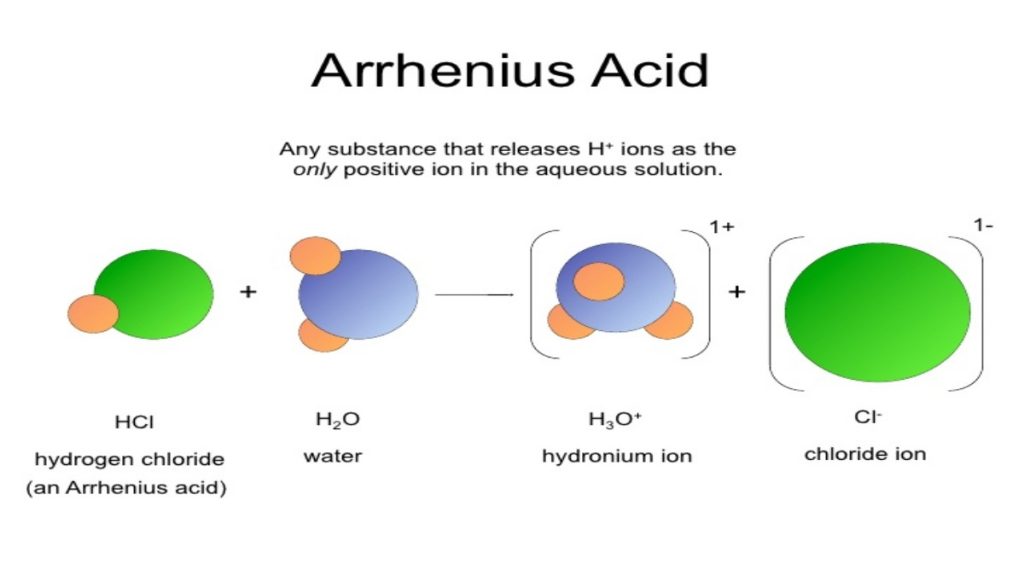
Arrhenius Acid
An Arrhenius acid is a molecule that when dissolved in water will donate an H+ in solution. Simply put, a proton donor.
The trick to recognizing an Arrhenius acid is to look for a molecule that starts with an H, and typically contains an oxygen or halogen.
Common examples of Arrhenius acids include:
§ Hydrochloric Acid – HCl
§ Nitric Acid – HNO3
§ Sulfuric Acid – H2SO4
§ Acetic Acid – HCH3CO2
§ and so many more…
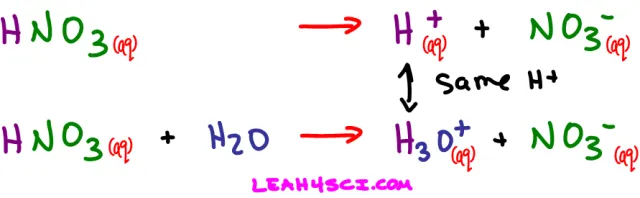
An acid dissociating in water does not form a free-floating proton. Instead one of the water molecules in solution will grab the H+ yielding a hydronium or H3O+ ion. Here’s what happens when nitric acid dissociates in water.
Arrhenius Base
An Arrhenius base is a molecule that when dissolved in water will break down to yield an OH- or hydroxide in solution. To recognize the Arrhenius base look for a molecule ending in OH, but not following CHx which refers to an alcohol.
Arrhenius base examples include:
§ Sodium hydroxide – NaOH
§ Potassium hydroxide – KOH
§ Magnesium hydroxide – Mg(OH)2
§ and so many more…

But what if the acid/base is not dissolved in water?
The Arrhenius definition for acids and bases only refers to compounds dissolved in water. Does this mean that acids and bases cannot exist out of water? Not quite, that’s where the Bronsted-Lowry definition comes in.
Bronsted-Lowry Acid
A Bronsted-Lowry acid, like an Arrhenius acid, is a compound that breaks down to give an H+ in solution. The only difference is that the solution does not have to be water. We can still refer to the exact same acids as listed for the Arrhenius acid examples, but this time we’ll change the solvent to ammonia, alcohol, or anything else.
We saw what happens when nitric acid (HNO3) dissolves in water. Now let’s see what happens when it dissolves in ammonia (NH3) or even methanol (CH3OH)
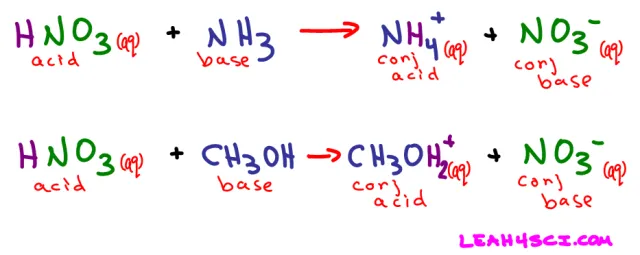
Nitric acid still dissolved to yield an H+ and NO3-, but this time it was NH3 and not water that picked up the free-floating proton.
Bronsted-Lowry Base
This is where we start to see the difference between the Bronsted-Lowry and Arrhenius definitions. While the Arrhenius base referred specifically to the hydroxide (OH-) ion, the Bronsted-Lowry base refers to any atom or ion capable of accepting or bonding to a free proton in solution.
Referring back to the HNO3 + NH3 reaction above, when ammonia picks up the free H+ it acts as a proton-acceptor. NH3 is the Bronsted-Lowry base in this example.
Additional examples include:
§ Methanol – CH3OH
§ Formaldehyde – H2CO
§ And even water – H2O