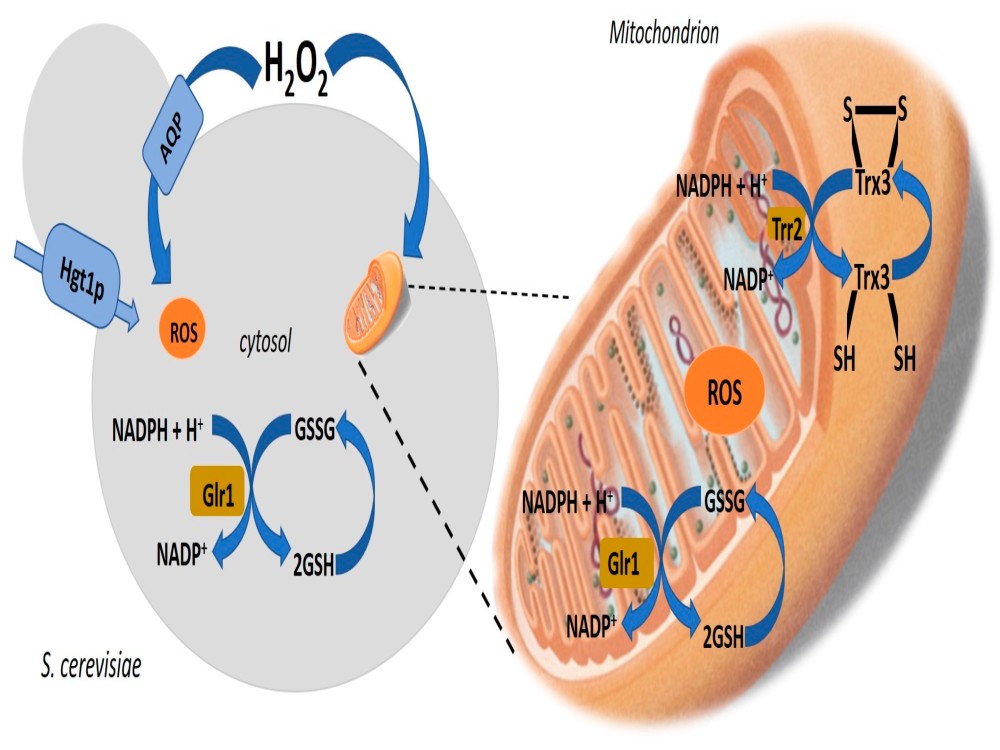
In aerobic respiration, the final electron acceptor is an oxygen molecule, O2. If aerobic respiration occurs, then ATP will be produced using the energy of the high-energy electrons carried by NADH or FADH2 to the electron transport chain. If aerobic respiration does not occur, NADH must be reoxidized to NAD+ for reuse as an electron carrier for glycolysis to continue. How is this done? Some living systems use an organic molecule as the final electron acceptor. Processes that use an organic molecule to regenerate NAD+ from NADH are collectively referred to as fermentation. In contrast, some living systems use an inorganic molecule as a final electron acceptor; both methods are a type of anaerobic cellular respiration. Anaerobic respiration enables organisms to convert energy for their use in the absence of oxygen.
Lactic Acid Fermentation
The fermentation method used by animals and some bacteria like those in yogurt is lactic acid fermentation (Figure 4.20). This occurs routinely in mammalian red blood cells and in skeletal muscle that has insufficient oxygen supply to allow aerobic respiration to continue (that is, in muscles used to the point of fatigue). In muscles, lactic acid produced by fermentation must be removed by the blood circulation and brought to the liver for further metabolism. The chemical reaction of lactic acid fermentation is the following:
Pyruvic acid +NADH↔lactic acid+NAD+Pyruvic acid +NADH↔lactic acid+NAD+
The enzyme that catalyzes this reaction is lactate dehydrogenase. The reaction can proceed in either direction, but the left-to-right reaction is inhibited by acidic conditions. This lactic acid build-up causes muscle stiffness and fatigue. Once the lactic acid has been removed from the muscle and is circulated to the liver, it can be converted back to pyruvic acid and further catabolized for energy.
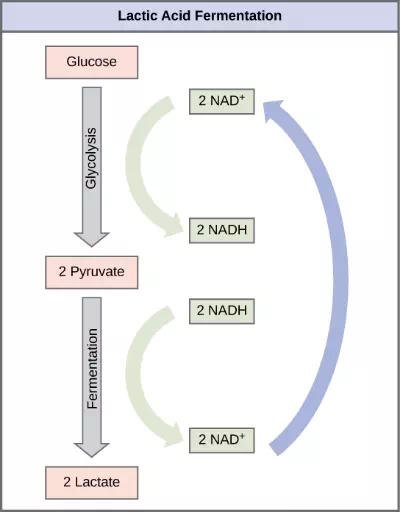
Figure 4.20
Tremetol, a metabolic poison found in white snake root plant, prevents the metabolism of lactate. When cows eat this plant, Tremetol is concentrated in the milk. Humans who consume the milk become ill. Symptoms of this disease, which include vomiting, abdominal pain, and tremors, become worse after exercise. Why do you think this is the case?
<!– The illness is caused by lactic acid build-up. Lactic acid levels rise after exercise, making the symptoms worse. Milk sickness is rare today, but was common in the Midwestern United States in the early 1800s. –>
Alcohol Fermentation
Another familiar fermentation process is alcohol fermentation (Figure 4.21), which produces ethanol, an alcohol. The alcohol fermentation reaction is the following:
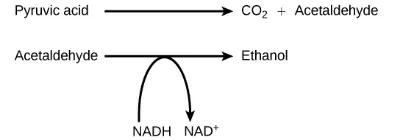
Figure 4.21 The reaction resulting in alcohol fermentation is shown.
In the first reaction, a carboxyl group is removed from pyruvic acid, releasing carbon dioxide as a gas. The loss of carbon dioxide reduces the molecule by one carbon atom, making acetaldehyde. The second reaction removes an electron from NADH, forming NAD+ and producing ethanol from the acetaldehyde, which accepts the electron. The fermentation of pyruvic acid by yeast produces the ethanol found in alcoholic beverages (Figure 4.22). If the carbon dioxide produced by the reaction is not vented from the fermentation chamber, for example in beer and sparkling wines, it remains dissolved in the medium until the pressure is released. Ethanol above 12 percent is toxic to yeast, so natural levels of alcohol in wine occur at a maximum of 12 percent.

Figure 4.22 Fermentation of grape juice to make wine produces CO2 as a byproduct. Fermentation tanks have valves so that pressure inside the tanks can be released.